 The element is in the 2nd column of the p block, Group IVA (Column 13). Germainum is in the 4th row Energy Level of the periodic table. Compared with potassium and sodium, rubidium is relatively rare, with a crustal abundance of just 78 mg kg-1 (Fyfe 1999). The d orbitals Groups 3-12 (columns) can hold 10 electrons.Įach energy level must be filled before moving up an energy level.Įach orbital group must fill before moving to the next orbital group. Rubidium-cesium-antimony coating is commonly applied to the photo-cathodes of photo-multiplier tubes, which are used in radiation detection devices, medical imaging equipment, and night-vision devices. The p orbitals Groups 13 - 18 (columns) can hold 6 electrons The s orbitals Groups 1 & 2 (columns) can hold 2 electrons The superscript tells us the number of electrons in the orbital. Diagram of the nuclear composition, electron configuration, and valence orbitals of an atom of caesium-133 (atomic number: 55), the most common isotope of this. The Coefficient tells us the Energy Level (Row) of the periodic table The electron configuration for the first 10 elements The "f block" on the periodic table are the Lanthanide and Actinide series.Įlectron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. The "d block" on the periodic table are groups 3-12 make up the d block and the elements' electron configurations end in d. The "p block" on the periodic table are groups 13-18 and end in p1, etc. The "s block" on the periodic table are groups 1 and 2 they end in s1 and s2. These rare earth metals are 2 periods behind because the f electrons are even higher in energy than the d electrons. The transition metals are behind by one period because the d electrons are high in energy.įor the rare earth elements (the Lanthanides and Actinides), they end in f. Scandium would end in 3d1, titanium in 3d2, etc. The general rule is that the element's electron configuration ends in d and whatever place they are in. And so it goes.įor the transition metals, groups 3-12, there are many exceptions. In group 4A or 14, all elements end in p2. Cesium là mt nguyên t hóa hc c biu th bng ký hiu Cs và có s hiu nguyên t là 55. Group 3A, or 13 all end their electron configurations in p1. Group 2 elements (2A), the alkaline earth metals, all end in s2 What period the element is in determines the 1st number.Įxample: H ends in 1s1 (even though H is not a metal, it resides in this group because it also has one valence electron) Group 1A (1), the alkali metals all end is s1. Hund’s rule of maximum multiplicity: The Hund’s rule of maximum multiplicity states that when several orbitals of equal energy are available, the electrons first fill all the orbitals singly before pairing in any of these orbitals.When looking at electron configuration, your fill order of electrons is: electron configuration: 1s2 2s2 2p6 3s2 3p6. Write the symbol and full electron configuration for the most likely monatomic ion formed from Ca. Pauli’s exclusion principle: The Pauli’s exclusion principle states that the two electrons in an atom cannot have the same set of all four quantum numbers. Write the condensed electron configurations for Cs. Other rules that explain the electronic configuration are as follows: Thus, the electronic configuration of cerium having atomic number 58 is as follows: The maximum number of electrons that can be accommodated is s-orbital are 2, p-orbital are 6, d-orbital are 10 and f-orbital is 14. 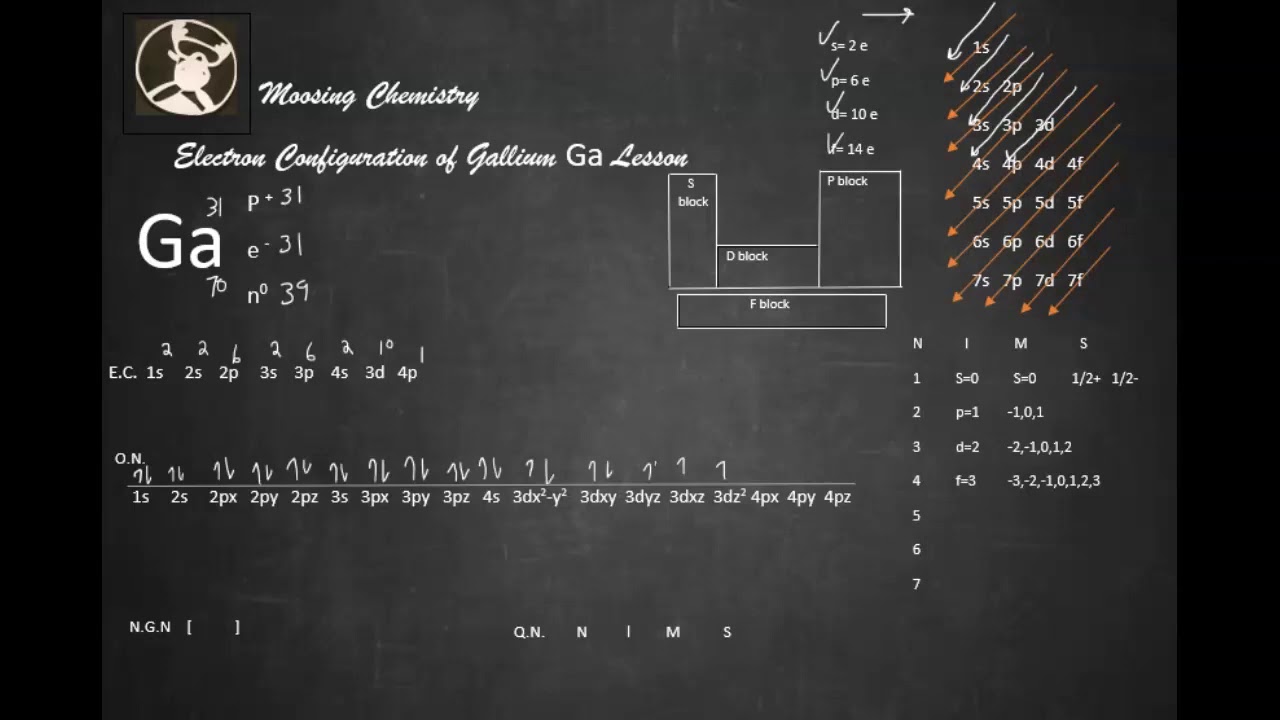 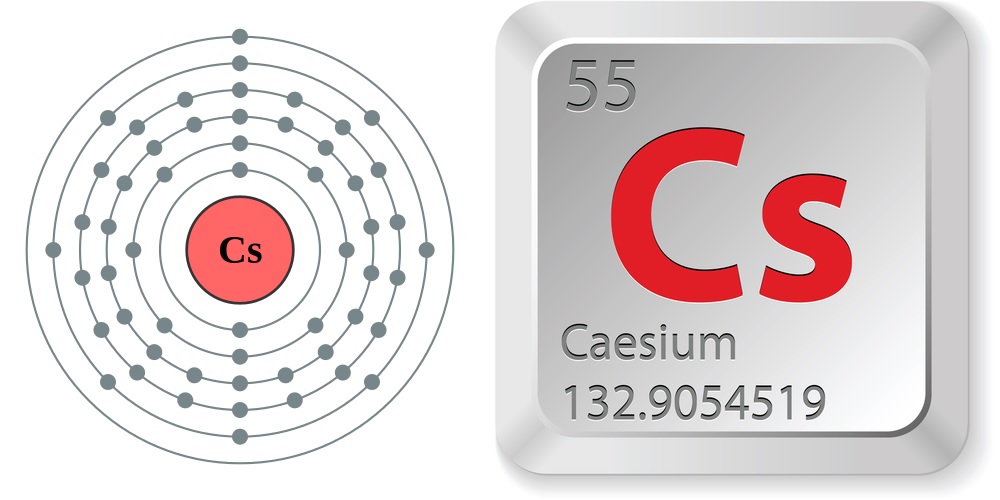
The order of energy of different orbitals in an atom is as follows: The Aufbau’s principle states that in the ground state of the atoms, the orbitals are filled with electrons in order of the increasing energies. The electrons fill up in the energy levels according to the Aufbau’s principle. Hint: The arrangement of electrons in the energy levels around the nucleus of an atom is known as the electronic configuration. Cesium, chemical element of Group 1 (also called Group Ia) of the periodic table, the alkali metal group, and the first element to be discovered spectroscopically (1860), by German scientists Robert Bunsen and Gustav Kirchhoff. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
